Compared to total body irradiation (TBI) or chemotherapy, antibody-drug conjugate (ADC) transplant conditioning may offer a more targeted and less toxic approach to clearing bone marrow niches to permit robust hematopoietic stem progenitor cell (HSPC) engraftment. Targeting the CD45 antigen, expressed on both HSPCs and immune cells, is of great interest for conditioning prior to both HSPC gene therapies and allo-transplantation, due to potential mitigation of immune rejection of neo or allo antigens. In this study, we investigated hematopoietic reconstitution and clonal dynamics following CD45-ADC (MGTA-45) conditioning and autologous transplantation of rhesus macaque HSPCs transduced with a barcoded lentiviral vector expressing the strong immunogen marker gene copGFP (GFP).
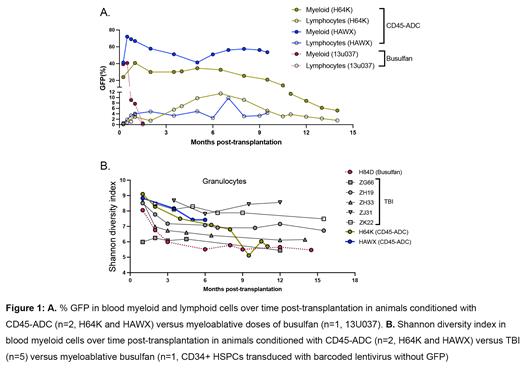
Two macaques had mobilized HSPCs transduced with the barcoded GFP-expressing lentiviral vector, and then received a single dose of 0.2mg/kg CD45-ADC as sole pre-infusion conditioning. On day 10 following CD45-ADC, transduced autologous HSPCs were thawed and reinfused. Both animals developed expected cytopenias, requiring transient platelet and red blood cell transfusion support, but no other toxicities were observed. Engraftment defined as recovery of neutrophil counts to >500/µL and no further need for transfusions occurred promptly by day 13 post-transplantation for both animals. Both showed stable high level multilineage engraftment with GFP-expressing barcoded hematopoietic cells, at levels equivalent to or higher than previously achieved with busulfan chemotherapy (Fig. 1A). Myeloid cells showed higher initial marking levels in both animals than T lymphocytes, with a slow increase in marked T lymphocytes over time, likely due to the interval required for development of naïve T cells from HSPC in an adult thymus. Barcode clonal tracking in both animals revealed highly polyclonal and diverse HSPC engraftment, with lineage-restricted and transient clones contributing for the first 1-2 months (m), replaced by contributions from multilineage stable clones, similar to clonal dynamics we have reported following TBI or busulfan conditioning in the same macaque lentiviral barcoding model (Koelle et al, Blood, 2017; Abraham et al, MTCD, 2023). Clonal diversity was high by Shannon index (Fig. 1B), with very high cumulative clone numbers per animal of 28-34,000, comparable to prior animals conditioned with TBI or busulfan. Beginning at 8m post-transplantation in the first animal (H64K) and 4m in the second (HAWX), clonal diversity began to gradually drop (Fig. 1B), with loss of 63% in first monkey (11m compared to 4m) and 34% in second monkey (6m compared to 3.5m) of detected clones in granulocytes, coincident with a reduction in the fraction of GFP-expressing cells in all lineages and also in vector copy number as assessed by ddPCR, suggesting potential rejection of cells expressing xenogeneic copGFP. Anti-CopGFP antibodies were detected in the serum of both animals, arising coincident with decreases in circulating GFP-expressing cells. Flow cytometric phenotyping of serial samples demonstrated only partial depletion of blood and lymph node T cells at the 0.2mg/kg dose of CD45-ADC, despite potent marrow HSPC depletion, and may account for the immune rejection of cells expressing a foreign protein. However, the rejection of GFP-expressing cells was much slower and less complete than following busulfan conditioning, when autologous HSPCs labeled with the same vector were rejected completely by 4-6 weeks post transplantation, suggesting some degree of tolerance induced by CD45-ADC conditioning.
Though long-term follow-up is needed to accurately compare ADC conditioning to TBI and busulfan, these findings underscore the potential of CD45-ADC conditioning as an efficacious, targeted and non-toxic approach to opening the bone marrow niche for genetically modified HSPCs. We are currently escalating the dose of CD45-ADC to achieve more profound T cell depletion at the time of transplantation, in hopes of producing lasting tolerance to vector-delivered transgenes or allogeneic HSPCs.
Disclosures
Palchaudhuri:Magenta Therapeutics: Ended employment in the past 24 months. Lanieri:Magenta Therapeutics: Ended employment in the past 24 months. Bhat:Magenta Therapeutics: Ended employment in the past 24 months. Tate:Magenta Therapeutics: Ended employment in the past 24 months. Guo:Magenta Therapeutics: Ended employment in the past 24 months. Olson:Magenta Therapeutics: Ended employment in the past 24 months.